World's most expensive drugs and two-year timer: why we hear more about spinal muscular atrophy
How Ukrainian parents fight for the lives of their children, why EU countries provide treatment at the budget expense, and our state holds off on the issue for more than one year, and what to do next, find out in our article.

SMA, spinal muscular atrophy, is a rare genetic disease. According to world statistics, this disease affects one or two babies out of 10,000 newborns. Every 40th planet's inhabitant, regardless of gender, nationality, and age, is a carrier of the "broken" gene that causes SMA. If two carriers meet, the probability of giving birth to a sick child is 25%.
Spinal muscular atrophy is called the number one genetic killer of children under two in the world. This orphan disease belongs to a group of fatal inherited diseases that destroy the motor neurons of the spinal cord. They're orphan because it's one of the diseases that are so rare and severe that for a long time, until recent years, pharmacological companies haven't invested in the development of drugs for patients with such diseases. So these diseases have long been abandoned, like orphans, not accepted by pharmaceuticals. Among the orphan diseases, there were widespread diseases such as tuberculosis, malaria, typhus, cholera because they were common in third world countries, and Western pharmaceutical companies were also not interested in drug development.
In Ukraine, the issue of young patients diagnosed with spinal muscular atrophy is acute: while abroad, children with the disease receive extremely expensive treatment at the expense of the state, in Ukraine, they are left derelict, leading to high mortality. We analyzed why it's happening and what can be done.
What is SMA?
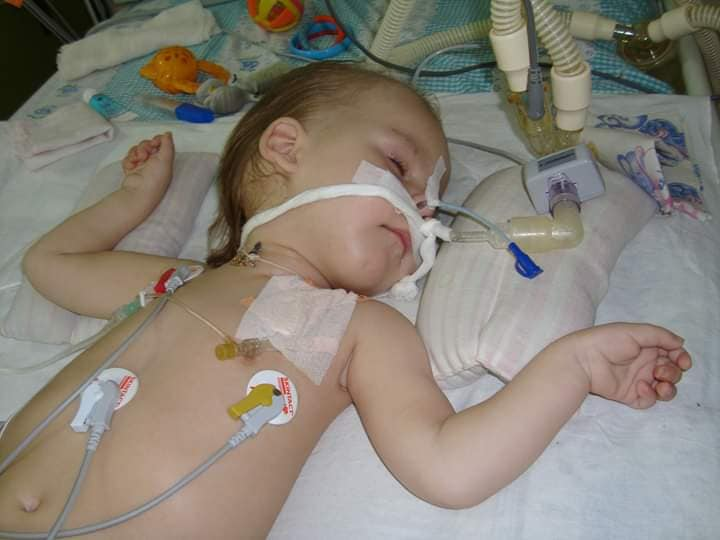
There are four types of muscle atrophy. More than half of babies diagnosed with type 1 don't live to see their second birthday. The disease affects all muscles without exception, including those responsible for swallowing, holding the head, motor functions, and breathing. And mental abilities don't suffer at all. Each day of little SMA kids means depending on a respirator, problems with eating, internal organ disorders, the constant need for care, maintenance and rehabilitation procedures, special equipment, and medicines. And understanding how your own body gradually refuses to work.
World's most expensive medicine
Until recently, there was no treatment for such children. But in the last five years, three drugs have appeared that can stop the disease.
The first is Spinraza, registered in 2016 by the pharmaceutical company Biogen (USA). Drugs don't repair the broken SMN1 gene but stimulate neighboring SMN2 to produce a full-fledged protein instead of the defective one. With regular use, Spinraza suspends the development of atrophy and stabilizes the patient's condition. Spinraza is used for injection. In the first year of treatment, a patient with SMA should make six injections into the spinal cord, then three injections per year. The patient should receive Spinraza all his life. In the summer of 2020, another drug for lifelong use would appear from Roche with the trade name Evrysdi. Evrysdi (active ingredient Risdiplan) increases and maintains the level of SMN protein; it's made as a syrup that should be drunk every day, and, unlike Spinraza, is suitable for all patients with SMA, even those who already irreversibly curved spine and injections are impossible.
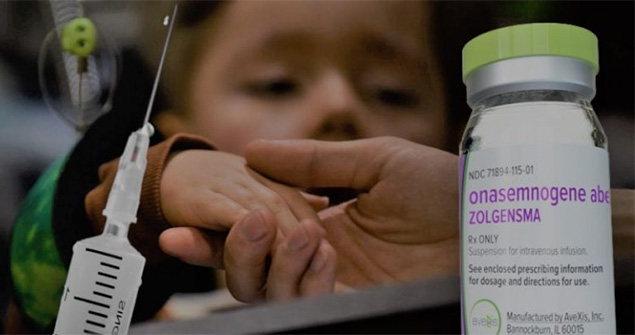
But before that, in December 2019, a genuine miracle happened. The Swiss pharmaceutical company Novartis presented the revolutionary development of the world's first gene therapy drug to treat SMA, Zolgensma. According to the manufacturer, these drugs can repair the defective SMN1 gene, and what's most impressive is that just one injection is enough to completely stop the disease.
Treating spinal muscular atrophy is extremely expensive, the cost of Spinraza is UAH 1,887,252, and Eurisdi is UAH 221,037 per bottle (annual cost depends on weight). The unaffordable price was considered fair because there aren't so many SMA patients, and drug development is extremely expensive. But the producers of Solgensma have estimated human life at an even more impressive amount, a single injection, which gives an SMA kid hope for a full life, costs 2.125 million dollars. Dozens of Ukrainian parents are now raising money for this drug on many online platforms and social networks. Today, the only way for children with SMA to recover from an extremely expensive injection is the generosity of those who care.
"Now the stories of SMA patients have become visible thanks to publications and reposts on social media," Lidia Babych, a Kyiv pediatrician and founder of the Pediatrics with Love Center said. "It seems that such children's numbers have suddenly increased. But it's not the case. In fact, these families finally have hope for their children's recovery. Scientists invented drugs that can completely cure a deadly disease. Forever. With just one injection of the drug.
But as always, there are a few various 'BUTS':
- drugs are intended for patients weighing up to 13.5 kg (according to other data, up to 21 kg);
- drugs don't restore those irreversible changes that have already developed;
- the drug is intended only for children under 2 years;
- it's the most expensive medicine on earth. More than $2 million per dose.
That is, the Zolgensma drug can cure infants of the most severe and deadly form of the disease, SMA type 1 and 2. But almost no one can pay for treatment and travel abroad on their own. And while fundraising continues, the child grows and 2-year restriction for administering a Zolgensma drug sounds like a sentence.
There are two more drugs, i.e. Spinraza and Evrysdi. These drugs are administered repeatedly, and the total cost of treatment is also extremely expensive. Last year, thanks to activists, they were finally registered in Ukraine. It's a very important step because even if there were funds, it was almost impossible to receive treatment for SMA in our country legally. Many parents also had to pay for traveling abroad for treatment. If you imagine a small child under the age of 2, who's chained to a bed and needs breathing equipment, tube feeding, has a serious condition, transporting such a baby abroad is a very complex and expensive process.
What about the state?
Unfortunately, the participation of the Ukrainian authorities in the fate of SMA kids has so far been limited to one registration of two SMA drugs, Spinraza and Evrysdi. At present in Ukraine, none of the SMA forms receives adequate support, treatment, or at least high-quality support testing from the state. Parents complain we don't even understand how to work with such children at all! There is no official count of children with this disease. Rehabilitation specialists don't know how to properly provide maintenance therapy. You won't even find food for children who are no longer able to eat on their own in Ukraine. Parents of SMA kids have to collect not only crazy money for treatment but also all the information about this disease on the Internet, while the child goes weak in front of the eyes. And if the lifeless "state machine" has the time and inspiration for a slow start and rolling, the SMA doesn't stop for a minute.
In the EU and the US, the state provides access to medicines to its citizens. In most European countries, Spinraza, the first drug to appear on the market, is already part of the reimbursement program.
You don't have to go far. In neighboring Poland, all patients with SMA are provided with these drugs at the expense of the budget. According to the Polish SMA Foundation, the practical stage in providing Poles with Spinraza began on December 17, 2018, when the Minister of Health of Poland Lukasz Szymanski and the Biogen company management, which produces Spinraza, agreed that these drugs would be covered by the public procurement program to treat all SMA patients "regardless of the form of the disease and the patient's age." The agreement was soon supported by a relevant order of the Ministry, and from January 1, 2019, the instrument was included in the list of drugs purchased from the budget.
Then a program was launched, during which they concluded contracts in Polish provinces between provincial branches of the National Health Fund and about a dozen local clinics. The next step was the mandatory tender procedure for the delivery of drugs to clinics and other logistical and organizational measures. Two or three months later, the drug reached the first patients. There are currently about 12-13 specialized centers in Poland where patients can receive Spinraza treatment. Specialists conduct tests to identify patients with SMA. All drug ampoules are strictly controlled, their injection is entered into the electronic system. It's called the "electronic monitoring system of programmed medication."
As Marcin Czech, President of the Polish Society of Pharmacoeconomics (ISPOR Poland Chapter), said in an interview with Interfax-Ukraine:
"For our country, it was, among other things, a political decision, because a large migration of Polish parents to other countries was happening: in search of treatment for their children, they went to France, Germany, Great Britain. We wanted to return them by providing treatment in Poland."
Since in Poland, the treatment of SMA is carried out at the expense of the state budget, they recently decided at the state level to introduce the screening of all newborns for SMA. Thus, now in Poland, there's not only treatment for SMA but also a program for screening children, which allows detecting the disease before the onset of symptoms. The screening program is only a part of the system, currently being developed in a pilot phase in several areas. But an entire complex of medical support works in Poland now. There are centers where you can get medical advice. The comprehensive patient support program also includes rehabilitation, special equipment, and, in part, social support from the state for parents and children.
The European Association SMA Europe also agrees with the need to introduce screening for all newborns. In August 2020, it announced the launch of a new European Alliance for Newborn Screening in Spinal Muscular Atrophy. The Alliance's main objectives are to reduce the time required to diagnose a child born with spinal muscular atrophy with the help of the NBS and help patient protection teams accelerate the detection of such children, given that early diagnosis and treatment of spinal muscular atrophy lead to better results. The founding members of this alliance are 19 national patient SMA organizations, members of SMA Europe, EURORDIS-Rare Diseases Europe, European Alliance of Neuromuscular Disorders Associations, TREAT-NMD, and pharmaceutical companies AveXis, Biogen, and Roche.
One of the SMA Europe members is the Ukrainian Charitable Foundation "Children with Spinal Muscular Atrophy," founded by parents of children with this orphan disease. It's the only Ukrainian organization that is a member of the international SMA community, recognized outside Ukraine. Among the Charitable Foundation partners are also TREAT NMD, Eurordis, EURO NMD network in ERNs. The Foundation has been conducting educational and legislative activities since 2004, funding research, providing comprehensive support to patients with SMA in Ukraine, and maintaining a register of patients (of which there are now about 250 in Ukraine, according to the Foundation).
Invisible children
The fund's president Vitalii Matiushenko lives in Kharkiv. The former physicist-scientist left his job to protect the right to life of his own daughter Yulia, who was diagnosed with SMA in the first year of life, and other SMA children.
For many years, Vitalii has been fighting the state. In civilized countries, it looks like a dialogue, but, according to the president of the Children with SMA Foundation, it's impossible to establish communication if one of the parties doesn't support this communication.
"If the state only declares that its priorities are the life and health of every person and doesn't confirm it with actions, then no desire for communication on our part will help here. In our country, orphan patients aren't liked, and SMA children have been completely removed from view. As if they don't exist."
For the last two years, the foundation has been trying to provide patients with SMA with free drugs necessary for life, i.e. Spinraza and Evrysdi. "All the technical aspects and elements that accompany access to treatment, have already been allegedly resolved: registration of drugs, analytical reports, research of the number of patients, all this is provided and discussed. A year ago, the 'Concept for the development of a system for assisting persons suffering from rare (orphan) diseases' was developed. But back in December 2020, the Ministry of Health held a meeting of a permanent working group, which includes drugs in the list funded by the budget, and what we see: drugs for patients with SMA weren't included in the nomenclature, although we created a roadmap. In Ukraine, the patient's role, as a sheep's, is to wait. While in other countries, the Ministry of Health already uses the experience of the patient as a full participant in the process of treatment."
The fact is that one of the registered drugs, Evrysdi, was available to Ukrainian patients through Roche's pre-registration access program, and about 50 patients in Ukraine received the drug for free, but now, in just a couple of months, the program will be discontinued. It's unknown what will happen to the patients who could receive the healing syrup before, but it's apparent that until Ukraine doesn't act further, they'll have to pay out of pocket not only for Spinraza but also for Evrysdi. For most patients, hundreds of thousands of dollars a year are unrealistic. This means that new deaths are inevitable.
In March this year, another petition for budget allocations for Spinraza and Evrysdi received more than 25,000 votes. The President's response soon appeared, stating: "I've instructed the Prime Minister of Ukraine, Denys Shmyhal, to take immediate measures to adopt an appropriate strategy for state support for patients with rare (orphan) diseases; to ensure systematic adaptation of the legislation of Ukraine to the needs of providing care to patients (clinical treatment protocols, clinical guidelines, etc.); to introduce the most optimal mechanisms for purchasing medicines to address the issue of providing SMA patients with effective treatment." That is, again, no specifics: they decided that something needs to be decided.
"I see corruption as a reason for a delay on the part of the state," Vitalii Matyushenko said. "Why did the entire journey from medicine to decision-making take a few months in that same Poland, and our country, more than a year has passed since the first drug was registered? We're facing new obstacles, while children continue to die. The Ministry of Health says that there is currently no practice and experience in creating such negotiation procedures. The question arises: why does it not happen with other drugs? Somehow these mechanisms still work, other nosologists receive drugs in the old way. We see systemic inhibition of the SMA."
Is there a solution?
What to do? Obviously, we need to put pressure on the authorities: the pressure is that there are few such children, and the cost of treatment for parents is sky-high. The state must provide these drugs, it's obliged! And according to Vitalii Matiushenko, society should focus not on promoting Solgensma, for doses of which volunteers are now actively raising money, but on the promotion of reliable drugs with proven action, like Spinraza and Evrysdi. He explains that parents' idea that their child will receive a single lifesaver may be misleading. About 200 people have used this drug worldwide during Solgensma's existence, and there's no data on what happens to such patients in five to six years. Besides, while millions are being gathered, a precious time is running out when children with SMA could receive treatment with other drugs. The so-called "golden window of opportunity," the time from diagnosis to the first symptoms of the disease, is rapidly closing. Parents wanting to receive a miraculous innovative tool can be understood, but Vitalii considers fundraising unacceptable because they distort the concept of the disease itself, and its treatment, and the state's attitude to such patients. It's the official position of the fund created and headed by him.
There's no single EU policy on providing all drugs to treat SMA. But today the full state supply of the Spinraza drug for all categories and different ages of patients is provided by EU members: Austria, Belgium, Germany, Luxembourg, Poland, Romania, and the Czech Republic.
"It's the experience of Poland and other EU countries that we should focus on," says Vitalii Matiushenko. "All objections that treatment is extremely expensive, that there are many patients in Ukraine (for comparison, Poland has almost four more of them), that it's a huge burden on the budget, and that they'll have to spend the budget on SMA only, are crushed by the example of neighboring countries. For example, Romania cannot be called a rich country either, but patients there still have treatment at the expense of the budget."
Public awareness of this issue, cooperation of medical and patient organizations, involvement of the press, public figures, and charitable organizations will be able to move the case and remind inapt Ukrainian officials what their responsibility is, and give SMA children a chance at life.
Who needs help right now?
Currently, several fundraising campaigns are underway in the Ukrainian information space for a precious injection of Zolgensma. There's very little time left for the "golden window of opportunity" for kids.
🙏 Dmytro Svychynskyi, Odesa. Dmytro is 4 months, SMA type 1. His father, Vitalii Svychynskyi, is raising funds. There are a little less than two months left for Dima to raise funds for the injection before severe symptoms appear: Dima's parents launched a flash mob on social media under the hashtag #DimaWe'llMakeIt. To date, a third of the required amount has been collected: UAH 21,671,780. But there's still time to help a boy.
🙏 Arsen Borys from Skalat city, Ternopil region. Arsen has an SMA type 2, on April 9, he'll be one and a half years old. Arsen's mother is Nadia Borys. UAH 22,351,085 was collected.
🙏 Viktoria Kotsovska, Lviv, 11 months SMA 1-2 type. Vika is no longer sitting and holding her head. Doctors say that there's a chance for improvement.
🙏 Adelina Motenko, Kakhovka, Kherson region. Adelina is 9 months. She has SMA type 1. UAH 23,000 was collected. Adelia's mother Natalia Motenko leads the fundraising.
The amounts are sky-high, but parents don't give up. Moreover, the precedent of successful fundraising in our country is already there: at the end of last year, with the efforts of caring people, they managed to raise the necessary $2.300 million (the price for Ukraine, which also includes logistics costs) for another SMA girl, eight-month-old Maria Sakhno from the city of Tavriia, Kherson region. The girl's mother Daria Sakhno shares her joy that in February this year, Maria had already received an injection in one of the Ukrainian hospitals:
"Masha got a chance to live without devices. Masha got a chance to see the sea or even the ocean, she got a chance to see next winter and touch the snow with her hands, Masha will live, Masha will breathe, and I believe Masha will walk!!!"
Another success story of Ukrainians in a bid for the lives of their children was the case of a child named Yelisei. The boy's parents, Karina and Oleksandr Zelenskyi, also raised money around the world, and then a miracle happened: Yelisei won an injection of Zolgensma in the manufacturer's lottery. Thus, the company annually allocates 100 doses of the drug, after which they're distributed among patients whose doctors apply for the lottery.
Also, read 7 solutions that support children with cancer in Ukraine.